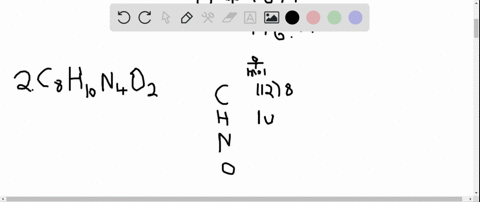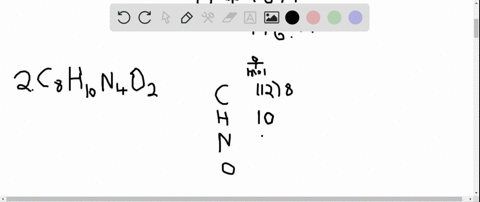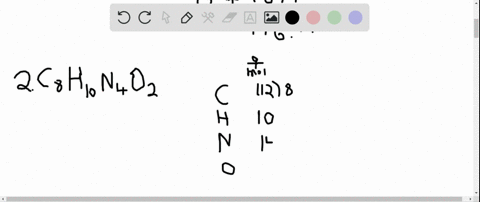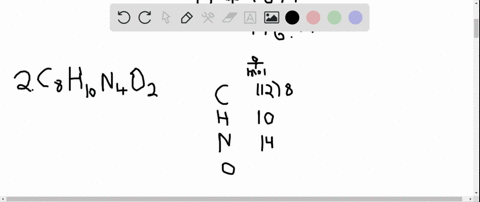
The answer must be rounded to three sig figs, the number of sig figs you have for the masses of the two reactants. Calculate the molar mass of water, which is two hydrogen atoms and one oxygen atoms molar masses combined: (2 × 1.008 g/mol) + 15.999 g/mol 18.015 g/mol. Iron is a metal, and there are many properties of iron like. #12.52 color(red)(cancel(color(black)("moles Fe"_2"O"_3))) * (color(blue)(2)color(white)(a)"moles Fe")/(1color(red)(cancel(color(black)("mole Fe"_2"O"_3)))) = "25.04 moles Fe"# So, the atomic mass or molar mass of iron will be the same and it is equal to 55.845 g/ mol. All you have to do here is to double the number of moles of iron(III) oxide and multiply the result by the molar mass of iron metal. they will not take part in the reaction.įor the last part, you got the approach down correctly. The rest of the moles of carbon monoxide will be in excess, i.e. 
#12.52 color(red)(cancel(color(black)("moles Fe"_2"O"_3))) * (color(red)(3)color(white)(a)"moles CO")/(1color(red)(cancel(color(black)("mole Fe"_2"O"_3)))) = "37.56 moles CO"# Step 1: Molar mass: The mass of 1 mole of a substance is called its molar mass. If all the moles of iron(III) were to take part in the reaction, then the reaction would also consume Right from the start, you should be able to tell that iron(III) oxide will be the limiting reagent because you have significantly more moles of carbon monoxide in #"2.00 kg"# than moles of iron(III) oxide in #"2.00 kg"#. In this case, iron (III) oxide has a molar mass of #"159.7 g mol"^(-1)# and carbon monoxide has a molar mass of #"28.01 g mol"^(-1)#. Iron III Oxide: Formula, Molar Mass, Uses & Solubility Iron II Oxide: Formula, Uses & Color Difference Between. When calculating molecular weight of a chemical compound. Finding molar mass starts with units of grams per mole (g/mol). 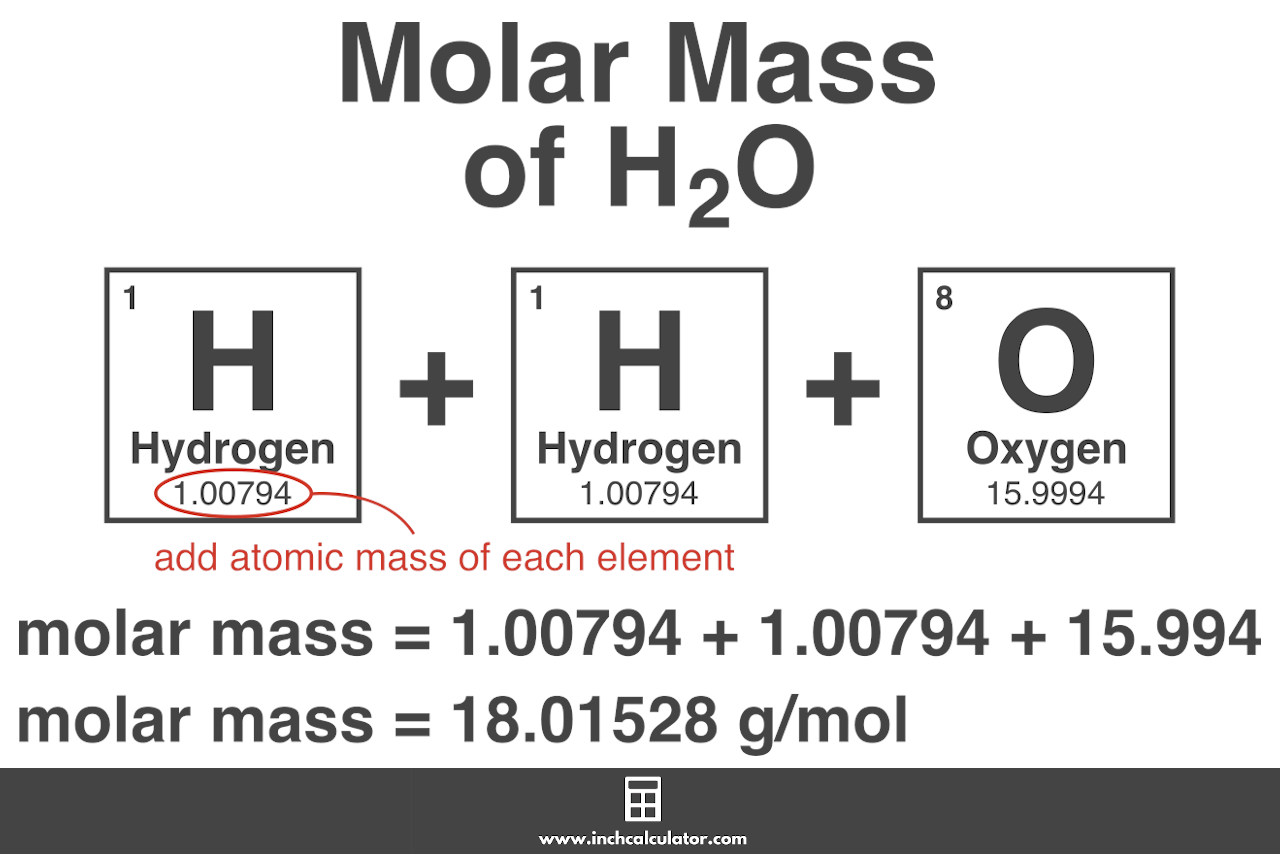
Similarly, since the atomic mass of iron is 56 u. When you're dealing with equal masses of two reactants, the reactant that has the greater molar mass will have the smallest number of moles present in the given mass. Convert grams Iron(III) Nitrate to moles. Please note that since the atomic mass of sodium is 23 u, the molar mass of sodium will be 23 g/mol. I'm not really sure I follow your calculations on this one.įor starters, a very quick way of figuring out which reactant will act as the limiting reagent when dealing with equal masses of the two reactants is to look at their molar masses. Molar mass is defined as the number of grams in one mole of a given element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
